Weicheng reported that a few days ago, Sichuan Baili Tianheng Pharmaceutical Co., Ltd. (hereinafter referred to as "Baili Tianheng", 688506.SH) released its 2024 annual report.
According to the data, in 2024, Baili Tianheng will achieve revenue of 5.823 billion yuan, a year-on-year increase of 936.31%; The net profit attributable to the parent company was 3.708 billion yuan, ending the situation of loss for three consecutive years.
As of the close of trading on April 3, Baili Tianheng's share price was 250.25 yuan per share, and the market value once again exceeded 100 billion yuan.
A record-breaking deal
Baili Tianheng's turnaround has to start with a transaction with a total transaction value of up to 8.4 billion US dollars. Because of this transaction, Chengdu pharmaceutical companies have become a new force to be reckoned with in the China Innovative Drug Club, which is dominated by entrepreneurs from Beijing and Shanghai.
On December 11, 2023, Baili Tianheng announced that it has reached a cooperation agreement with global pharmaceutical giant Bristol Myers Squibb ("BMS") for a self-developed anti-tumor drug molecule, BL-B01D1. The potential total transaction value of the collaboration is up to $8.4 billion, including an upfront payment of $800 million, up to $500 million in near-term contingent payments, and up to $7.1 billion in additional payments upon achievement of development, registration and sales milestones.
At the same time, Baili Tianheng has set a record for the largest licensing transaction volume in the field of innovative drugs in China to date, as well as a record for the total transaction price of a single product of ADC (antibody conjugate) drugs in the world.
With the arrival of the down payment of $800 million in March 2024, Baili Tianheng ended its three-year losing streak. According to the annual report, in 2024, Baili Tianheng will achieve revenue of 5.823 billion yuan, a year-on-year increase of 936.31%; The net profit attributable to the parent company was 3.708 billion yuan, compared with a loss of 780 million yuan last year.
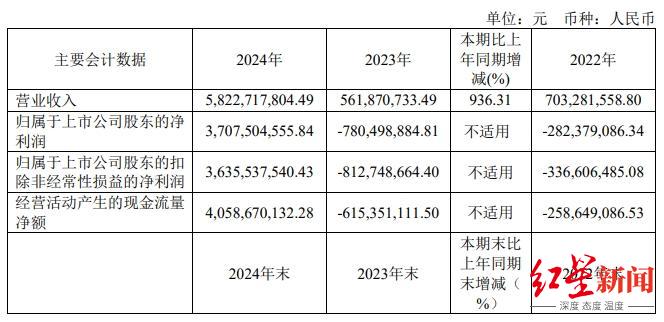
Baili Tianheng's 2024 revenue data, the source of Baili Tianheng's 2024 annual report
In addition, for Chinese pharmaceutical companies, the deal is a rare opportunity to secure a co-development model, unlike most of the previous days, when all interests were sold at once.
According to the agreement, the two parties will jointly develop and share future revenues.
The benefits to Baili Tianheng may be more profound than the maximum contract amount of $8.6 billion. The partnership also gives the company access to partners' global market commercial resources, including but not limited to an established distribution network, experienced sales resources, proprietary market insights, and more...... This will also help to promote the company's growth into a global pharmaceutical company (MNC) with a global leading edge in the field of oncology drugs in the future. Baili Tianheng said in the annual report.
From generics to innovative drugs
Zhu Yi, the founder, chairman and chief scientific officer of Baili Tianheng, led the team to negotiate with BMS, and he led the transformation of Baili Tianheng from generic drugs to innovative drugs.
Zhu Yi is from Neijiang, Sichuan, and in 1980, at the age of 17, he was admitted to Sichuan University to study radio physics. The radio major was suggested by his father, hoping that he would have a skill after graduation. Later, Zhu Yi discovered that he was really interested in biology, and in 1984, Zhu Yi was admitted to Fudan University to study for a master's degree in biophysics.
After graduating with a master's degree, Zhu Yi did research on microbial immunity in universities for a period of time. However, that was still far from the innovative breakthrough he craved. Later, he resigned to do foreign trade and real estate.
After earning the first pot of gold by relying on his real estate career, in 1996, Zhu Yi returned to Wenjiang, Chengdu to found Baili Pharmaceutical (the predecessor of Baili Tianheng) and returned to the pharmaceutical industry. Baili Pharmaceutical started with chemical generics and Chinese patent medicines, and the first generic product is the antiviral drug ribavirin granules (trade name: Xinbolin), which was launched in 1998.
In 2008, Baili Pharmaceutical acquired Sichuan Shule Pharmaceutical Co., Ltd. (hereinafter referred to as "Shule Pharmaceutical"), a local veteran state-owned pharmaceutical company. After the completion of the acquisition, the sales volume of the anesthetic drug "propofol" under Shule Pharmaceutical increased significantly, from hundreds of thousands a year to more than 10 million a year. In 2011, 2012 and 2013, propofol injection contributed 22.81%, 20.72% and 24.27% of the revenue of Baili Tianheng respectively, which was the largest proportion of revenue among all product categories.
By 2010, Baili Tianheng has built a complete R&D, production and marketing system, and its generic drug business can steadily bring tens of millions of "spare money" to the company every year. Zhu Yi believes that it is time to make innovative drugs.
The U.S. front-end discovery, China's back-end development
At the beginning of 2014, in the cold winter of the development of the global biotechnology industry, Baili Tianheng established SystImmune, an innovative drug research center in the United States, and later invested most of the revenue from the chemical preparation and Chinese patent medicine preparation business in the research and development of innovative drugs. Regarding the strategic layout of innovative drugs, Zhu Yi's idea is: front-end discovery in the United States and back-end development in China.
In the period of generic drugs, Baili Tianheng did chemical drugs and Chinese patent medicines, but when choosing the direction of innovative drugs, Zhu Yi aimed at the direction of anti-tumor macromolecule drugs in the field of biological drugs, including ADC, GNC, ARC.
Among them, ADC is a key modality for cancer treatment. According to the annual report, the global ADC market size will reach US$10.1 billion in 2023 and is expected to reach US$151.9 billion by 2033, and the share of ADCs in the entire oncology market is expected to increase from 4.5% to 26.4% during the same period.
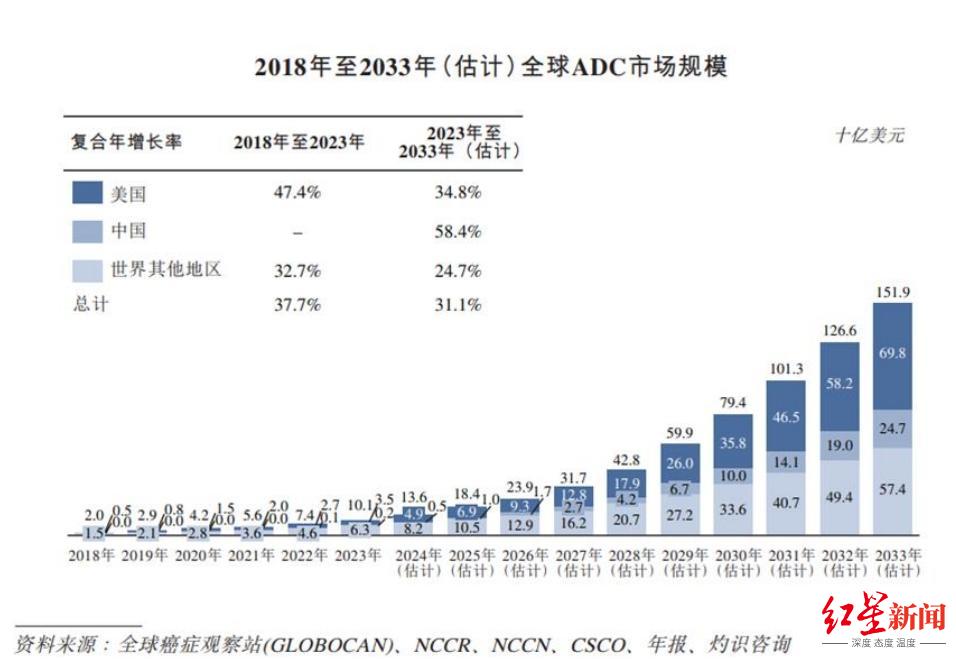
Source: Baili Tianheng 2024 annual report
BL-B01D1, the drug molecule favored by BMS, is the world's first-in-class, new concept and the only EGFR×HER3 bispecific antibody ADC to enter phase III clinical stage. At present, the clinical trials of BL-B01D1 have covered different tumor indications such as lung cancer, breast cancer, gastrointestinal cancer, urological cancer, gynecological cancer, and head and neck cancer, and has the potential to become a cornerstone drug for pan-tumor treatment.
For Baili Tianheng, the primary goal in the short term is to accelerate the clinical development of BL-B01D1 in China and globally. According to the prospectus of Baili Tianheng Hong Kong stocks, it is expected to submit the first indication new drug marketing application to the China Food and Drug Administration no later than 2026; The first BLA (Biologics License Application) will be submitted to the U.S. FDA as early as 2028. In the next three to five years, BL-B01D1 will submit regulatory approval applications for more indications in China, Europe, the United States and other regulatory regions.
Under the contract with BMS, the company will receive a non-refundable milestone payment of $250 million if, by the end of 2025, the first Phase II or Phase III trial of the Licensed Product as a first- or second-line treatment in the U.S., and a further non-refundable milestone payment of $250 million before the end of 2026, the first Phase III trial of the Licensed Product as first-line therapy in the United States.
This means that if all goes well, in the next two years, Baili Tianheng will receive $500 million.
Ticker Name
Percentage Change
Inclusion Date