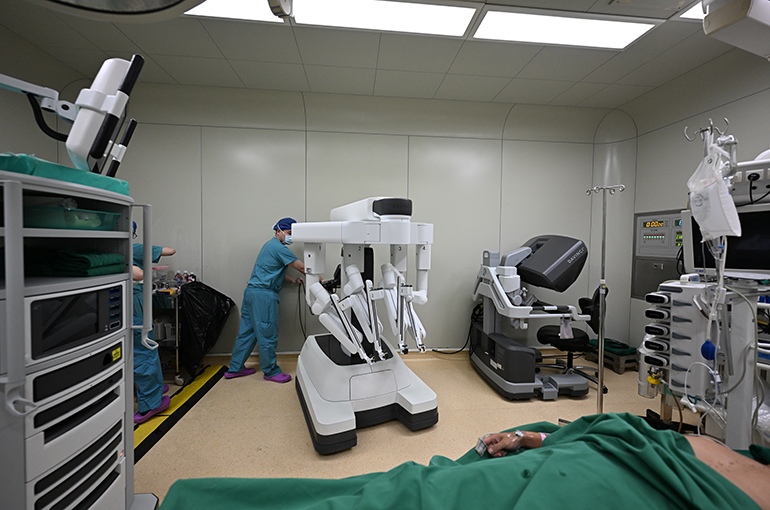
 Beijing Pushes Surgical Robots, Expands Policy Support for Innovative Drugs and Devices
Beijing Pushes Surgical Robots, Expands Policy Support for Innovative Drugs and Devices(Yicai) April 8 -- Beijing has encouraged promoting the application of new technologies such as surgical robots in hospitals in its latest annual industry guidance document for the development, market access, and clinical use of innovative drugs and medical devices.
The document, led by the Beijing Municipal Medical Insurance Bureau and jointly issued by 10 local government agencies yesterday, also highlighted the inclusion of eligible technologies in the pricing framework for medical products and supports their coverage under medical insurance payments.
Included were 32 measures, covering the research and development of pharmaceuticals and medical devices, clinical trials, evaluation and approval, production and distribution, and clinical application.
Beijing called for conducting clinical effectiveness evaluations and health tech assessments for the application of surgical robots. It also stipulated that eligible technologies should be included in pricing frameworks and supported by medical insurance payments to accelerate the use of multi-category surgical robots, including for laparoscopy, orthopedics, neurosurgery, and interventional procedures, in hospitals.
In addition, Beijing encouraged setting high-value medical equipment leasing platforms, including for robots, while promoting innovative leasing models for surgical robots to accelerate their iterative innovation. The document incorporated various factors into the performance monitoring of public hospitals, including collaborative R&D of surgical robots, the effectiveness of their use, and the openness of robotic applications across all scenarios.
Beijing will also provide financial support to companies developing innovative drugs and medical devices. The municipal government investment fund will collaborate with central and district-level government investment funds to invest over CNY10 billion (USD1.5 billion) in innovative pharmaceutical firms this year.
In addition, the Beijing government will fund no fewer than 100 early-stage innovative drug projects through fiscal appropriations, venture capital, loans, and others.
Editors: Tang Shihua, Martin Kadiev